Visualizing Clinical Trials
Whether you’re in pharma, biotech, or somewhere in between, there is a powerful moment of truth when your drug has gotten its IND approved, gone through animal trials, and is ready for human trials. There are a lot of new challenges involved. Clinical trials are where all of the big risks live, often costing over $2b per new drug or biologic.
Back in the old days, clinical trials were monitored entirely by site visits. All Interim Monitoring Visits (IMVs) were conducted in person by a Clinical Research Associate. As costs have risen and technology has progressed, sponsors have insisted on using a combination of Remote Monitoring Visits, Risk-Based, and Real-Time Monitoring.
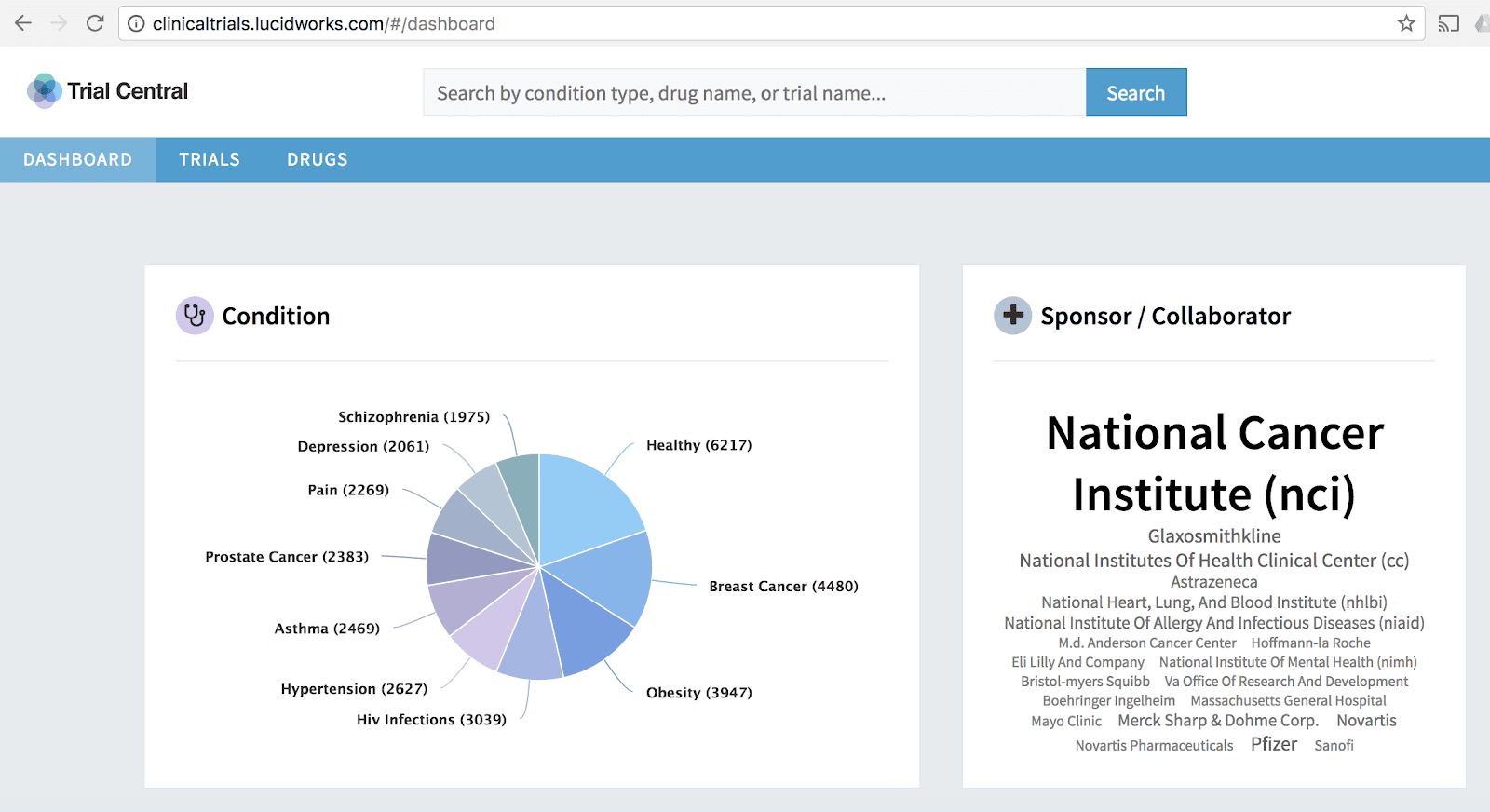
Research funded by the National Institutes of Health (NIH) shows that with better visualization, understanding clinical trials data required on average 28.1% less time while maintaining similar accuracy. Moreover, the research shows that this doesn’t change based on the participants familiarity of statistics or clinical trials procedure. According to the study, “the combination of having a visualization to reference while reading the status quo published report can further help to save time and increase accuracy.”

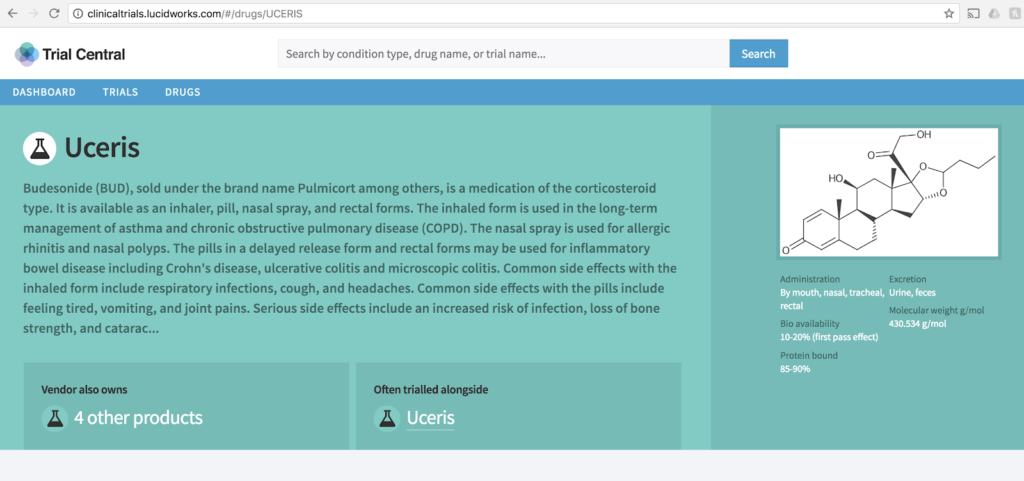
At Lucidworks we think you should both be able to visualize and explore clinical trials data. In our example we ingested data from ClinicalTrials.gov and applied our search and AppStudio visualization technologies to it. You can learn more in our Understanding Clinical Trials Data webinar.
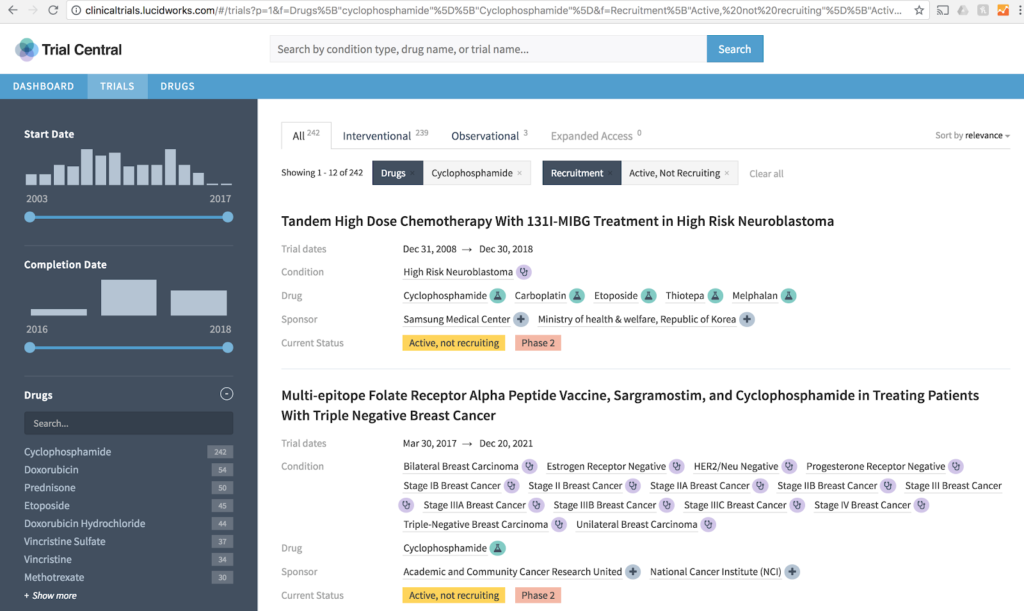
At Lucidworks, we think this holds true well beyond clinical trials data (and other research backs that). Whether in the Biotech industry, small molecule, or other life sciences, adding better search and visualization can make any data easier to explore and comprehend. This speaks to use cases throughout the research, development, commercialization and marketing lifecycles.
Learn more:
- Watch our Understanding Clinical Trials Data Webinar
- Contact us, we’d love to help!
LEARN MORE
Contact us today to learn how Lucidworks can help your team create powerful search and discovery applications for your customers and employees.